Energy Bands in Solids
Last modified 2020-01-07
Sini
The electrons in a free atom can only have discrete energy levels. An electron farthest from the nucleus has the highest energy. The farther the electrons are, less the energy required to escape from the electrical pull of the nucleus. When an electron absorbs heat or light, its energy increases. Electrons jump up to higher level orbits when acquiring energy and release energy when they jump down to original energy level.
Hydrogen Energy Level Plot
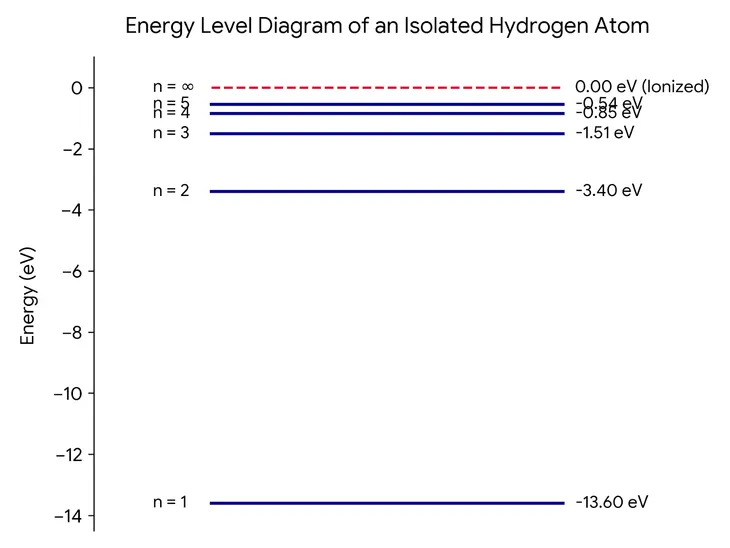
En = (-13.6/n2) * eV
where $n = 1, 2, 3...
Ground State (n = 1) is the lowest and most stable energy level. As we see above, the isolated hydrogen atom’s electrons have a single energy value depending on its energy state. But when atoms combine to form solids, this doesn’t stay true. If two isolated atoms are brought together, then electrons in orbits are influenced by the nucleus and electrons of the other atom. In a solid, every electron is influenced by the neighbouring atoms. Hence the energy level of two electrons in the same orbit can experience slightly different force on it, and the energy of two electrons can also be slightly different even if they occupy the same orbit. For the inner electrons which are bound closely to the nucleus of an atom, this change in energy levels is less, but the difference in energy level increases for outer shells.
If we plot the energy level of a single Si atom, we will get a plot similar to that of the hydrogen atom. In an Si crystal, each solid Si atom positions between four other Si, and each of these Si atoms share one electron with the central atom. Hence all the atoms form a covalent bond with four other Si atoms, completing the outermost shell. Since the valence band is now filled, no electron movement occurs, and the Si crystal cannot conduct electricity.
If we were to plot the energy level for solid silicon, we’ll get:
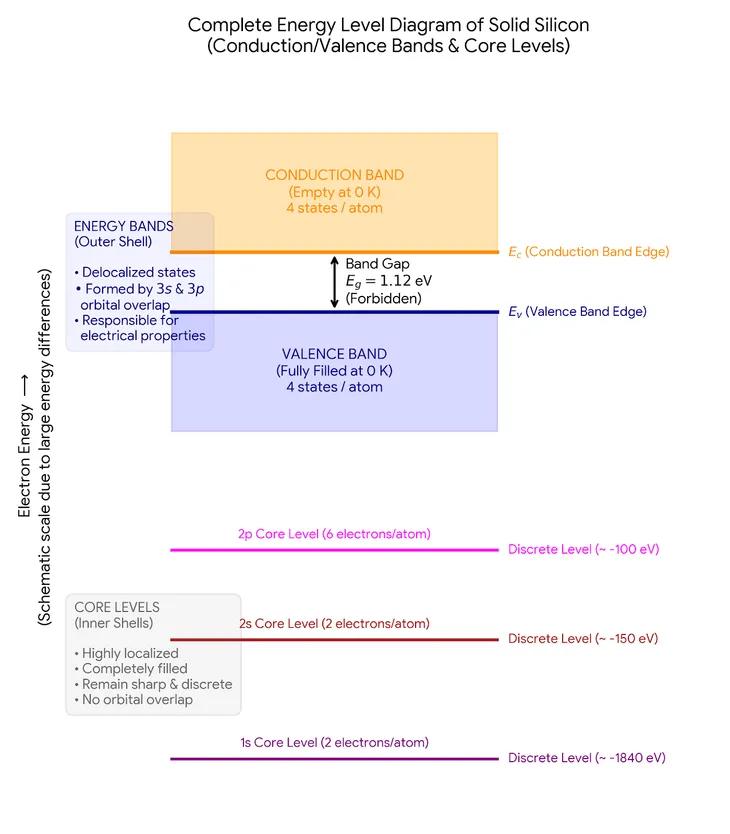
There is also a conduction band given in the picture above. If an electron in the third band can acquire enough energy, it can move to the next energy level band, which is called the conduction band. In this band, the electrons are freed free from the atom, and can move randomly along the crystal. When a material has such free electrons they can conduct electricity.
Next: Bandgap